It soars over the tossing waves on enormous, outspread wings. With nary a flap, the bird is soaring dynamically high above the ocean, its eyes scanning the sky around it and sea below. It may seem unremarkable to us today: the feet are webbed and extend behind it, the tail is short and blunt, the body rounded and muscular, but encased in a bullet-like shell of feathers, dark above and light below. The wings are mostly arm, the hand bones short. The feathers are short, and the arms make up most of the wing length. It looks like an albatross, but is several times larger than any living albatross; but it is otherwise unremarkable in its appearance from any other great seabird. That is, until you examine its head.
There are many types of ocean-going flyers, pelagic birds of one type or another. Those that hug the shores or feed in the shallows such as cormorants and auks, and those that make vast searches of the ocean such as boobies, and albatrosses. Examining their wings will tell you much about how they get around; and examining their jaws will tell you how they might catch their prey — together, these tell you what they might feed on, what they prefer.
Perspective
 120 odd million years ago, the first animals which might be called birds evolved. And regardless of what some few disgruntled scientists (and so far from the majority in both practice and theory as to be negligible were it not for that some creationist groups love to talk about them) may think, most evidence indicates that these first birds were certainly dinosaurs. Their ancestors were toothed, but all modern birds are toothless, and it is this transition that has fueled some debate in the last century. It had long been argued that the teeth of Archaeopteryx lithographica, that urvogel, were so like some reptiles (lizards) that the bird-dinosaur connection was impossible; or that the jaw of Ichthyornis dispar was almost a perfect match as a small mosasaur jaw and was comparable in most details. But it is clear that from the point that we recognize the beginnings of birds, teeth were there to stay. From Archaeopteryx to Ichthyornis, a fully-toothed jaw was present during the rise of birds. Some birds experimented: Some enantiornithines lost their teeth, but they were still present across a broad swath of the diverse group.
120 odd million years ago, the first animals which might be called birds evolved. And regardless of what some few disgruntled scientists (and so far from the majority in both practice and theory as to be negligible were it not for that some creationist groups love to talk about them) may think, most evidence indicates that these first birds were certainly dinosaurs. Their ancestors were toothed, but all modern birds are toothless, and it is this transition that has fueled some debate in the last century. It had long been argued that the teeth of Archaeopteryx lithographica, that urvogel, were so like some reptiles (lizards) that the bird-dinosaur connection was impossible; or that the jaw of Ichthyornis dispar was almost a perfect match as a small mosasaur jaw and was comparable in most details. But it is clear that from the point that we recognize the beginnings of birds, teeth were there to stay. From Archaeopteryx to Ichthyornis, a fully-toothed jaw was present during the rise of birds. Some birds experimented: Some enantiornithines lost their teeth, but they were still present across a broad swath of the diverse group.
Some birds lost only some teeth. Prior to the rise of the clade called Avialae, which includes Archaeopteryx but not dromaeosaurids like Velociraptor, teeth had been lost (or strongly reduced) multiple times in close relatives: the ornithomimosaurs show progressive loss from Harpymimus okladnikovi (toothed at the tips) to all other ornithomimosaurs; therizinosauroids went from fully toothed jaws to loosing teeth in their premaxillae (but keeping all the rest); and in oviraptorosaurs, the jaws of the basal members were short, peg-like, and included some giant premaxillary teeth (Incisivosaurus gauthieri), became smaller and lost in all bones but the premaxillae (Caudipteryx zoui), then were fully lost. This may have occurred convergently in some of these groups, with multiple losses.
This has been one of the more interesting questions of my research: Why? One attempt to answer this has been persistent in bird research for the last decade: Birds lost their teeth so that they could fly.
Taking Flight
But there are problems with this idea.
First, by the time birds became volant, were able to fly, had shrunk to the size of pigeons or jays, they still had teeth, and as in the case of tern-sized Ichthyornis, had retained enough teeth in their jaws to be confused with non-avian jaw bones. It didn’t matter that Archaeopteryx had less than 20 pairs of teeth per jaw, Ichthyornis had many more. Related to Ichthyornis was the diving, flightless bird Hesperornis regalis. This bird had developed vestigial wings, but massive legs and long, robust toes, reminding the describers who worked with it in the late 1800s and early 1900s of a loon and a grebe (both birds were capable of flight, however).
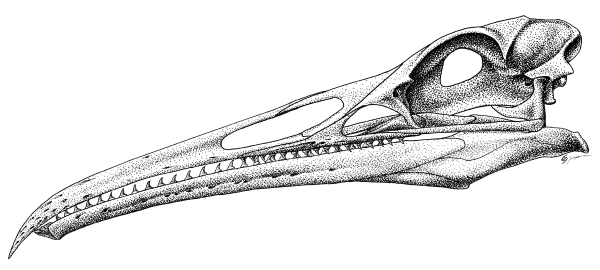 But the skull is another story. Like therizinosauroids (but convergently, mind you!) teeth were lost in the premaxilla, but are extensive on the lower jaw and in the maxilla. Yet Hesperornis, you might say, was without the ability to fly, so what purpose does discussing the teeth here serve? Because the same complement of teeth, and even that form of jaw, are present in Ichthyornis. A fully volant, ocean-going, flapping-winged, powered-flying bird.
But the skull is another story. Like therizinosauroids (but convergently, mind you!) teeth were lost in the premaxilla, but are extensive on the lower jaw and in the maxilla. Yet Hesperornis, you might say, was without the ability to fly, so what purpose does discussing the teeth here serve? Because the same complement of teeth, and even that form of jaw, are present in Ichthyornis. A fully volant, ocean-going, flapping-winged, powered-flying bird.

Ichthyornis dispar, from Marsh’s Odontornithes monograph (1880). Complete jaws, premaxillae, and fragments of the maxilla are know that reveal their concordance to the outline above, which shows a composite of related basal carinatan birds from the Niobrara Chalk (Clarke, 2004).
Hesperornis and Ichthyornis lay at the cross-roads of late-1800s thinking regarding the rise of birds and their evolution from whatever flightless form preceded them. Beyond them, we had Archaeopteryx, and modern birds. Here we had these clear intermediates, younger than the one and older than the other, with less toothed jaws and clearly “reptilian” mandibles. Birdy in every other way, they asked: What was so special about the jaw? Perhaps, it was presumed, birds could not fully fly until they started losing teeth?
The value of the tooth to flight so preoccupied some it was necessary for them to assume some special quality to their implantation and development of tooth germs for replacement as a way to explain this phenomenon. This got pulled into the debate on bird origins: Dinosaurs has these deeply recessed sockets for teeth, with all sorts of complex tissues, that meant the teeth involved such complicated mechanisms that merely losing them would never bring about a bird-like jaw. Yet lizards, for example, were more simple in their tooth-implantation, lacking true sockets, and could thus afford the less complicated tissues and thus loss. The deep socket was a problem! It has later been shown that well, no: birds with teeth (real birds, like Hesperornis above) have tooth sockets, like crocs and dinosaurs, and that the histology of these structures includes the presence of alveolar bone, a type of tissue that develops from the dental ligament that ossifies as a substance not too dissimilar to the osteosclerotic layering of bone inside long bones and the beaks of birds (and some pterosaurs!) that creates medullary bone. The walls between tooth sockets are made of this tissue, and they’re present in birds. Tooth sockets aren’t just deep divots on the tops of jaw bones, as might be the case in some acrodont lizards and other archosauromorphans like rhynchosaurs, but are remnants of a long dental groove into which teeth are embedded in this ligament that ossifies. Thus, the dental groove of ichthyosaurs becomes the precursor system for archosaur tooth implantation. Including birds.
Loss of teeth in birds was securely restricted to flight ability. Teeth must, therefore, be so much heavy in comparison, requiring such material! So long has it been assumed, that it is shocking to some that it’s been known over 50 years that the material elements that comprise teeth and the need to anchor them to the jaw (the development of additional interdental ligaments, alveolar bone and the medial bony dentigenous wall, of the teeth themselves be they mostly dentine or with enamel, and the additional soft-tissue structures needed to fuel these organs, in their development or in life) are in fact lighter than those of the beak. The cornified sheets that make up the bird’s beak overall cover more area, wrapping around the oral margin and the outside of the jaws, often forming secondary palatal and ant-palatal platforms on the mandible, and can be very thick (e.g., in hornbills and in parrots).
Cost, however, is another matter, as the tissues that form the beak arise as a development of the dermis: they are, essentially, grown from the skin and comprise the same keratin structures as do nails and hair, but the tissues are chemically somewhat different. Teeth also arise as dermal growths, but the buds that become teeth becomes isolated from the dermis early on, and different genes are involved. More tissue differentiation occurs in tooth development than beaks, and the organs of tooth anchorage and replacement are persistent in teeth, whereas the same tissue replacement in beaks are present that regrow lost skin are restricted generally to those tissues. The presence of teeth also involve the development of additional dermal structures, such as lips, etc., which are often excluded by the presence of beaks. In other words: Teeth are more expensive, but overall lighter, than they beaky counterparts, but only if you consider teeth alone; whereas if you consider all the other tissues that might be present, the absence of teeth might be “lighter.” But they may also be (much) more costly.
Why Loss?
So what actually supports the progressive loss of teeth. Well, the short answer may end up being that teeth are not that useful for some things. The need to keep them going is one thing, but their actual use is another. For one teeth, teeth are isolated structures that, when absent, make portions of the jaw less useful when biting some things. When present, they are useful when used along with the rest of their number. A row of steak knives in the jaw of Allosaurus or Carcharodontosaurus are useful in that each tooth in the row pulls at the flesh as the animal bites. Tiny denticles grip bits of skin and muscle and apply greater tension in the tissues around the tooth-bite, so when the jaw is pulled through flesh it all tears and comes with the tooth. Each tooth is a battalion making its way through the opposing host. Remove one, and the effect is lessened. Mechanically, if you want a tooth of this type, you need many. In the more croc-like morphology, teeth aren’t serrated but the biting is done along most of the jaw and is useful for catching prey that can be elusive, and thus may not be caught on one part of the jaw. Normally, it’s at the tip, but one cannot pick when the prey slips away. Without that ability to chose where you bite, the teeth become generalized and mostly the same size so they may be equally effective. Mechanically, the jaw is still more efficient at the tip and applies greatest force at the back, closer to the muscle. Losing teeth impairs the effectiveness of the teeth in other parts in these regions. So either you have them, and minimize effect of losing teeth (regrowing them) or you lose them (and develop alternatives).
One alternative is the toothless beak, and it is so useful that in archosaurs it has evolved multiple times: The crocodilian lineages include Macelognathus (=Hallopus?) vagans, Lotosaurus adentus, and the dinosauriform lineages include the tip of the dentary in some Silesauridae, completely in the basal ceratosaur (noasaurid?) Limusaurus inextricabilis, most of the jaw in Harpymimus okladnikovi and completely in all other ornithomimosaurs, the maxilla and lower jaw in Caudipteryx zoui, the tip of the lower jaw in Incisivosaurus gauthieri, and apparently completely in all other oviraptorosaurs where it may persist onto the palate itself, and multiple times in bird evolution: the confuciusornithids, some enantiornithines such as Longipteryx chaoyangensis (only the tips of both extremely elongated jaws toothed) in contrast to Gobipteryx minuta (completely toothless), and of course modern birds and their direct antecedents such as Apsaravis ukhaana. Interpolated in here were animals with fully-toothed jaws. But these animals all tend to lose teeth in some parts of the jaw first, and this is what might be termed a “clue.”
First, almost all toothless archosaurs lose teeth in their premaxillae and/or at the tips of their lower jaws. It’s not true in ornithomimosaurs and oviraptorosaurs, but this is hardly disconfirmation. This implies that the tips of the jaw are behaving somewhat differently than the rest of the jaw. Strains on the jaw may be more useful without teeth. Strain analysis, performed by multiple groups, on jaws with and without teeth suggest that the presence of teeth helps reduce strain in the tips of the snout, compared to toothlessness … unless there’s a beak on the tip. That is, a rhamphothecal sheath.
When the tip of the snout is put under strain, the forces acting on it seek to dissipate through the skull. Mostly, this occurs through various joints. a flat joint does a poor job of dissipation: strain goes into the connective tissues and those tissues bear more strain than the bones, and this can cause the bones to buckle against one another. However, if the bones interlock, including if they are fused, strain may dissipate across the joints and thus bear more of the strain. This tends not to happen in toothed jaws: strain is dissipated from the teeth into the connective tissues into the jaw bone, and the first point of weakness is the tooth/non-tooth interface. Teeth, being unfused to their jaw bones, may tend to break or be loosened, and strain can be dissipated. Teeth, as such, are useful ways to limit strain into the rest of the skull, and loss of a tooth is easily mitigated by the regrowth of another. Toothless jaws lack this, but that’s actually fine: strain in toothless jaws carries into the skull, and if the skull is partially fused, all to the good. Bird skulls are often almost fully fused, and strain dissipates from one end to the other.
Biting (vertical forces) act in different ways in different parts of the jaw. At the tips, these forces translate horizontally, and a curved tip helps mitigate possible shear while increasing efficiency, so predatory birds specializing on large prey items may typically have deep tips (raptors, owls, vultures, shrikes, etc.; comparably, predatory rails like phorusrachids develop hooked tips). The trick to curved tips is thus, in some ways, to mitigate strain elsewhere in the beak. Like a single large, giant tooth. A rhamphotheca acts as a sort of mitigation, in another way: when present, strain can be dissipated from bone into the skin, as it typically already does, but the harder rhamphotheca carries this force across its surface and if it is present over more extensive regions of the skull, strain may then dissipate across the face despite the presence of open joints in the skull. Developed cornified tissues such as sheets of this structure present in the facial dermis of some animals acts this way, as in musk oxen and bighorn sheep, whereas in some extinct animals similar structures would have acted to pull strain away from points of impact (pachycephalosaurs; Snively & Cox, 2008) and during prolonged agonistic “pushing contests” (some ceratopsians, tyrannosaurs? — Snively et al., 2006). But also during biting, fusion of some skull bones appears to coincide with development of a denser, cornified tissue over the skull (Snively et al., 2006), and may help adapt the more sledgehammer-mode of head strikes in some other theropod dinosaurs, such as abelisaurs.
If Not Teeth, Then…?
Loosing teeth, then, seems like a bit of a benefit, but it also carries an additional effect. A smooth, straight margin of the jaw makes for poor handling of prey items. The jaw margins are inefficient for resisting strain of a struggling animal, and this may be mitigated through increased bite forces (i.e., stronger jaw muscles, increased leverage) but these aren’t available in most birds: most of the jaw muscles are pretty small, and when they are large the beak is also short and deep (e.g., parrots). That other effect, then, is the loss of advantage apart from the jaw tip. So some birds have developed unusual gripping strategies. Excluding use of feet, birds can develop ridges on their palates, formed of cornified plates or other skin attributes, of cornified tongue structures, or soft, cornified palatal ridges, protrusions of the bones themselves, and other, more interesting modifications. Penguins, for example, are fast-swimming pursuit predators and thus often have little time or ease to grab prey, so they ram feed: open mouth, swallow prey as it comes within the jaws. To facilitate this grip, penguins (like some turtles) have unusual projections of the esophageal lining that form cornified spikes, and so do some ducks.
 The diversity of anseriform beaks is particularly intriguing. Ducks, unlike what has been reported, do not have simple smooth beaks in their mouths, but various combinations of ridges, tubercles, projections, and even serrations of the jaw margin itself. Mergansers, unlike most ducks, lack the wide “duckbill” that so characterizes swans, geese, ducks proper, screamers, etc. Instead, mergansers have very narrow, pointed jaws. And they have serrated bony edges. These edges underlie on a one to one basis a larger, pointier serration of the rhamphotheca, and these serrations extend from the back end of the upper beak to the near the hooked tip. There are marginal serrations on the lower jaw as well, but they do correspond to bony projections. But it demonstrates a living bird which developed serrated jaw margins. And these serrations are useful for gripping prey, such as small slippery fish. They’re pointed back into the mouth, and concentrated underneath the external bony nostril (or naris) where they are also largest, implying this is the point of greatest mechanical utility.
The diversity of anseriform beaks is particularly intriguing. Ducks, unlike what has been reported, do not have simple smooth beaks in their mouths, but various combinations of ridges, tubercles, projections, and even serrations of the jaw margin itself. Mergansers, unlike most ducks, lack the wide “duckbill” that so characterizes swans, geese, ducks proper, screamers, etc. Instead, mergansers have very narrow, pointed jaws. And they have serrated bony edges. These edges underlie on a one to one basis a larger, pointier serration of the rhamphotheca, and these serrations extend from the back end of the upper beak to the near the hooked tip. There are marginal serrations on the lower jaw as well, but they do correspond to bony projections. But it demonstrates a living bird which developed serrated jaw margins. And these serrations are useful for gripping prey, such as small slippery fish. They’re pointed back into the mouth, and concentrated underneath the external bony nostril (or naris) where they are also largest, implying this is the point of greatest mechanical utility.
Whilst not living, a larger, ocean-going group of birds one-upped the merganser. While considered tentatively associated with Anseriformes, Pelagornithidae are a group of gigantic, volant soaring birds (with the lowest-aspect wings of all birds known) formerly associated with gull-like boobies and other pelicaniform birds. Now extinct, these birds evolved during the Paleogene and persisted into the Pleistocene. Human foragers venturing towards the oceans likely encountered these birds. Pelagornithids look like gulls, but aren’t procellariiforms; rather, various structures of their palate and skull indicate a relationship with geese, swans, and ducks, but perhaps also with fowl (Galliformes). It seems peculiar that the anseriform radiation has produced a number of awesome, gigantic clades, including the “Demon Duck of Doom” — also known as mihirungs — Dromornithidae, giant Pacific island moa-nalo (the “turtle-jawed geese,” which are actually giant anatines and thus “true ducks,” including Thambetochen), and Gastornithidae, which includes the infamous “horse-eater” (it wasn’t, this is a paleo meme) Diatryma. Peculiarly, like mergansers and the turtle-jawed ducks, the margins of the beak in pelagnornithids are serrated, and this indicates that the rhamphotheca that covers them would be serrated, too.

Skull and jaws of Pelagornis sandersi Kspeka (2014). While based on a crushed specimen, the skull and jaws of related species (such as Pelagornis mauretanicus) are useful in creating a sort of composite.
What need would a bird have for these? Investigations have occurred into the microstructure of pelagnornithid “pseudoteeth”: Hildegarde Howard described in 1957 the possible ecology and beak structure of peleganornithids, presuming that the bony-tooth projections of the jaw margin must have been covered in rhamphotheca, a topic further supported by microhistological work from Antoine Louchart and colleagues in 2013 that indicates that a rhamphotheca must have existed because the surfaces of the projections were covered by extensive very soft tissue and were only somewhat mineralized. Extensive force mitigation must not have occurred, and this may suggest that, like sperm whales, pelagornithids generally specialized in soft-bodied prey (cephalopods like squid or jellyfish) often found near the ocean’s surface. These projections thus do not compare well with teeth, or the “toothed” jaws of mergansers and moa-nalos. This soft-body prey capture hypothesis is also aided by the extreme size variability in the jaws: only some projections are very long, and the interlocking of such things may vary; the lower jaw passed inward of the upper, and long channels on the palate would have accomodated the tips of the longest pseudoteeth. Growth of the projections and their rhamphothecal covering were covered by Louchart et al. (2013), but they were unable to come to a conclusion regarding how the rhamphotheca would develop over the projections in time. Juveniles may have lacked them, and developed them as the rhamphotheca was developing, remaining soft as in kiwis and some ducks, but hardening quickly when they became self-sufficient foragers.
Birds losing teeth is a transition many lineages took, but some adaptive advantage allowed a serrated margin to develop and persist in some extinct and living lineages. Toucans, some hornbills, even some hummingbirds, today bear slight or large serrations, all of which are wholly made of keratin, whereas the turtle ducks and mergansers developed marginal bony serrations exaggerated by the beak itself, and pelagornithids took this to eleven. These birds vary in what they feed on, from vegetation, fruits, with some omnivorous capabilities. Hummingbirds, for example, while nectarivorous, also consume insects while on the wing (serrations tend not be advantageous for the smooth, hard, chitinous exosekeletons of arthropods). But the development of “toothed” birds, exemplified below with both actual and “fake” teeth …
 … shows that there have been many adaptive developments of similar structures, but that their functions can reveal distinctions in the result. This may explain why some pelagornithids have more recurved pseudoteeth, whereas others have forward-projecting ones. A visual similarity to teeth does not necessarily imply a mechanical correlation. This also leads us to consider a purely visual component to these structures, but the study to separate this visual component from a feeding one is difficult in extinct birds, and were it found to be true in those, it may not be true in extinct forms. Thus the nature of these structures, found so far in extinct forms exclusive of teeth (so far! “Wait for it!” he says cryptically) implies developmental needs to retain serrated jaws.
… shows that there have been many adaptive developments of similar structures, but that their functions can reveal distinctions in the result. This may explain why some pelagornithids have more recurved pseudoteeth, whereas others have forward-projecting ones. A visual similarity to teeth does not necessarily imply a mechanical correlation. This also leads us to consider a purely visual component to these structures, but the study to separate this visual component from a feeding one is difficult in extinct birds, and were it found to be true in those, it may not be true in extinct forms. Thus the nature of these structures, found so far in extinct forms exclusive of teeth (so far! “Wait for it!” he says cryptically) implies developmental needs to retain serrated jaws.
(Incidentally, we’re not talking about some recent efforts to create living monsters in the form of growing tooth germs on the jaws of Gallus embryos in order to hatch toothed chickens for the sake of some need to have sabretoothed chickens or whatever. No, we’re not talking about it; I certainly am not.)
Clarke, J.A. 2004. Morphology, phylogenetic taxonomy, and systematics of Ichthyornis and Apatornis (Avialae: Ornithurae). Bulletin of the American Museum of Natural History 286: 1-179.
Howard, H. 1957. A gigantic “toothed” marine bird from the Miocene of California. Bulletin of the Department of Geology of the Santa Barbara Museum of Natural History 1: 1-23.
Ksepka, D. T. 2014. Flight performance of the largest volant bird. Proceedings of the National Academy of Sciences of the United States of America 111 (29): 106204-10629.
Louchart, A., Sire, J.-Y., Mourer-Chauviré, C., Geraads, D., Viriot, L. & de Buffrénil, V. 2013. Structure and growth pattern of psuedoteeth in Pelagnornis mauretanicus (Aves, Odontopterygiformes, Pelagornithidae). PLoS ONE 8 (11): e80372.
Marsh, O. C. 1880. Odontornithes: A monograph on the extinct toothed birds of North America. (United States Geological Exploration of the 40th Parallel, U. S. Government Printing Office, Washington, D. C.) [Available online, OA, here at Archive.org; PDF link here]
Snively, E. & Cox, A. 2008. Structural mechanics of pachycephalosaur crania permitted head-butting behavior. Palaeontologica Electronica 11 (1): 3A.
Snively, E., Henderson, D. M. & Phillips, D. S. 2006. Fused and vaulted nasals of tyrannosaurid dinosaurs: Implications for cranial strength and feeding mechanics. Acta Palaeontologica Polonica 51 (2): 435-454. [PDF]





“some enantiornithines such as Boluochia zhengi (toothed mandible and a deep, toothless premaxilla)”
That was the original interpretation of Boluochia, but O’Connor et al. (2011: http://www.tandfonline.com/doi/abs/10.1080/14772019.2010.512614#.VE9N3hZGXh8) found that the “deep shape” of Boluochia premaxilla is an artefact and that the premaxilla bears teeth, as in other longipterygids (that bear toothed premaxilla and rostral dentary and lack teeth in maxilla and dentary mid- and posterior thirds).
I will fix this posthaste.
Great post. Gotta love all the crazy things animals do with soft tissue. I’ve wondered for a while now if piscivorous spinosaurids had tongue or roof of mouth tubercles as many fish eating birds do…
This is actually something I would put in the plausible category only if there was no other easy prehension mechanic for the animal. As it is, it’s only penguins, ducks, turtles, related forms, with these structures, and they all lack any other form of prehension other than the jaws. Spinosaurids have forearms with grappling/snaring capability, not to mention feet, to aid in prey restraint, not to mention rendering of prey into parts, so they’d likely not need methods such as throat spines in order to prevent prey, swallowed whole and alive, from wriggling free.
I think it’s fairly obvious that tooth loss isn’t correlated to flight capacity. In their 160 million years of fully capable aeronautic capacities, pterosaurs only lost their teeth more or less 5 times – far less so than the number of times birds and their non-avian dinosaur cousins became toothless -, while bats are yet to develop platypus-like beaks.
The idea that teeth were lost for flight comes from people who were only familiar with mammals. Mammal teeth are heavy. I once got to handle a sheep skull and was surprised to find that the center of gravity was between the molars, below the palate.
Bats probably can’t evolve a beak. Therians in general probably can’t, because that would apparently require a caruncle (a keratinous analog of the egg tooth) to start from. Monotremes retain such a thing, marsupials have an extremely reduced version, and placentals lack it altogether.
I think since mammals can produce some of the necessary dermal tissues involved, it is possible, but perhaps not likely, that these tissues can encroach on the face. If so, they can develop to the point that they might cover the oral region. I don’t think there’s a possibility of this, though, simply because the complex of facial muscles are always present in all mammals. If anything, reduction in facial muscle would occur only when the muscles couldn’t function as effectively, and that only seems to happen when the rostral teeth become super large, as in mole roles, in which case teeth take over the “beak” role. So yea, when it comes to a beak replacing teeth, I doubt it on two grounds: It’s far more likely, as with some rodents, to merely reduce the teeth in favor of a select set (incisors) or produce cornified skin of the lips and palate (well, most mamals have cornified skin on their palates/tongue, but externally I am thinking of some edentates).
Yes, the evolution of cornified beaks in therians is likely constrained by the requirement of functional facial musculature so helpful in sucking from the mum’s mammillary papillae. Monotremes in contrast possess no mammillary papillae, milk is licked by the hatchlings directly from mum’s body surface (or hairs, as far I know), so they were free to experiment with beaks and other facial extravagances (like proboscis, etc).